Pfizer COVID-19 vaccine for children approved in Canada
Posted November 19, 2021 4:00 am.
Last Updated November 19, 2021 3:49 pm.
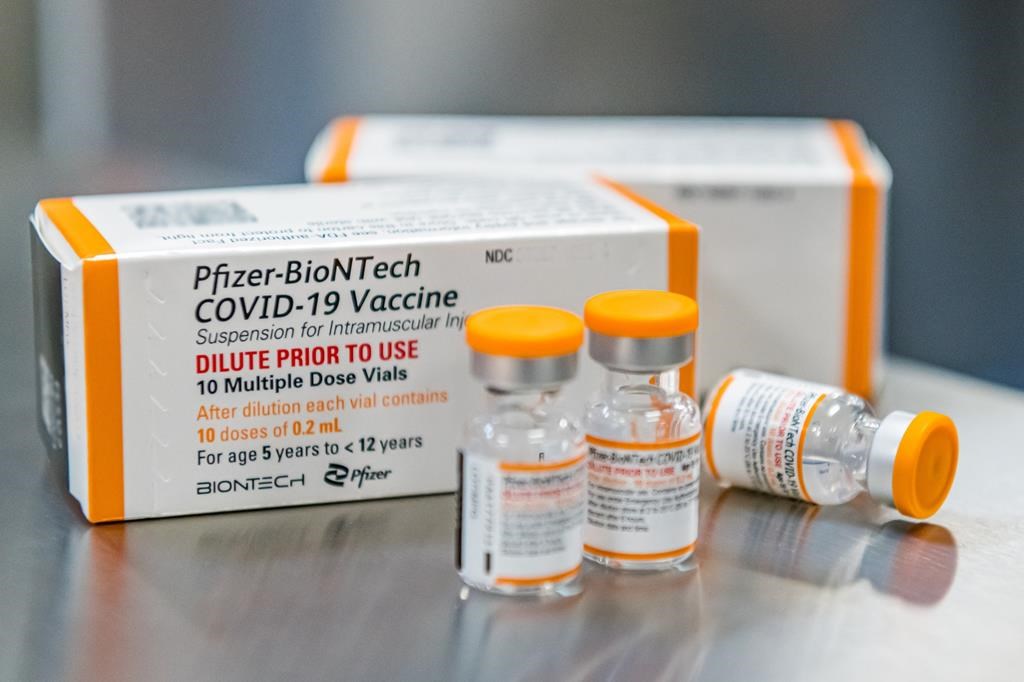
Health Canada has authorized the use of the Pfizer COVID-19 vaccine in children aged five to 11.
Canada is expecting an accelerated delivery of 2.9 million child-sized doses of the vaccine, enough for a first dose for every child in the five to 11 age group.
BREAKING: The Procurement Minister says Canada will start receiving the Pfizer shots for 5-11 year olds on Sunday. All 2.9-million doses will arrive by the end of the week #cdnpoli
— Cormac Mac Sweeney (@cmaconthehill) November 19, 2021
Pfizer and its partner BioNTech say the results of their trials in children show comparable safety and efficacy results to those recorded in a previous study in adults aged 16 to 25.
“After a thorough and independent scientific review of the evidence, the Department has determined that the benefits of this vaccine for children between 5 and 11 years of age outweigh the risks,” officials said in a statement released Friday.
The kids’ vaccine was approved with a three-week interval between doses, but the National Advisory Committee on Immunization has suggested at least eight weeks between doses. Evidence has shown that a longer interval between doses increases the efficacy of the drug, and may even reduce the risk of rare side-effects like inflammation of the heart.
NACI’s advice is that children who do not have any conditions that would make vaccination risky may receive two doses.
The language is slightly less strong than that for 12 to 16-year-olds, who should receive the vaccine according to NACI.
The recommendation for younger kids will likely be strengthened over time, as more information becomes available, said Dr. Matthew Tunis, NACI executive secretary.
Clinical trial showed that the immune response in children 5 to 11 years of age was comparable to the immune response in people 16 to 25 years of age. The vaccine was 90.7 per cent effective at preventing COVID-19 in children 5 to 11 years of age and no serious side effects were identified.
Main side-effects for children associated with the vaccine are the same as those for slightly older vaccine recipients, though they were less common in kids, aside from redness and swelling at the injection site.
While children are at a lower risk of severe outcomes related to COVID-19, some kids do get very sick, chief public health officer Dr. Theresa Tam said.
Officials also point to cases of multisystem inflammatory disease, long-COVID, and heart inflammation associated with COVID-19 among kids as reasons to get the kids the shot.
“Most of the outbreaks now that are reported to us are from the school and daycare setting and especially in primary school,” Tam said. “So we know that school outbreaks and disruption to education has a big impact on children as well.”
For now NACI suggests children wait at least 14 days before or after receiving another vaccine, such as the flu shot, to get the COVID-19 vaccine so that officials can more easily identify potential side-effects.
Health Canada is still reviewing Moderna’s mRNA COVID-19 vaccine for children aged six to 11 years of age.
The U.S. Food and Drug Administration approved the Pfizer-BioNTech vaccine for children on Oct. 29, and the United States has already vaccinated more than two million children.
Dr. Supriya Sharma, Health Canada’s chief medical adviser, said no safety issues have emerged during the U.S. rollout of the vaccine.








